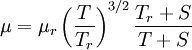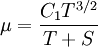Sutherland's law
From CFD-Wiki
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | In 1893 [http://en.wikipedia.org/wiki/William_Sutherland_(physicist) William Sutherland], an Australian physicist, published a relationship between the absolute temperature, <math>T</math>, of an ideal gas and its dynamic visocity, <math>\mu</math>, based on | + | In 1893 [http://en.wikipedia.org/wiki/William_Sutherland_(physicist) William Sutherland], an Australian physicist, published a relationship between the absolute temperature, <math>T</math>, of an ideal gas and its dynamic visocity, <math>\mu</math>, based on kinetic theory of ideal gases and an idealized intermolecular-force potential. This formula, often called Sutherland's law, is still commonly used and most often gives fairly accurate results with an error less than a few percent over a wide range of temperatures. Sutherland's law can be expressed as: |
| - | :<math>\mu = \ | + | :<math>\mu = \mu_r \left( \frac{T}{T_r} \right)^{3/2}\frac{T_r + S}{T + S}</math> |
| + | |||
| + | :<math>T_r</math> is a reference temperature. | ||
| + | :<math>\mu_r</math> is the viscosity at the <math>T_r</math> reference temperature | ||
| + | :S is the Sutherland temperature | ||
Some authors instead express Sutherland's law in the following form: | Some authors instead express Sutherland's law in the following form: | ||
| Line 9: | Line 13: | ||
Comparing the formulas above the <math>C_1</math> constant can be written as: | Comparing the formulas above the <math>C_1</math> constant can be written as: | ||
| - | :<math>C_1 = \frac{\ | + | :<math>C_1 = \frac{\mu_r}{T_r^{3/2}}(T_r + S)</math> |
== References == | == References == | ||
* {{reference-paper|author=Sutherland, W.|year=1893|title=The viscosity of gases and molecular force|rest=Philosophical Magazine, S. 5, 36, pp. 507-531 (1893)}} | * {{reference-paper|author=Sutherland, W.|year=1893|title=The viscosity of gases and molecular force|rest=Philosophical Magazine, S. 5, 36, pp. 507-531 (1893)}} | ||
Revision as of 13:46, 17 May 2007
In 1893 William Sutherland, an Australian physicist, published a relationship between the absolute temperature, 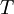 , of an ideal gas and its dynamic visocity,
, of an ideal gas and its dynamic visocity, 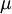 , based on kinetic theory of ideal gases and an idealized intermolecular-force potential. This formula, often called Sutherland's law, is still commonly used and most often gives fairly accurate results with an error less than a few percent over a wide range of temperatures. Sutherland's law can be expressed as:
, based on kinetic theory of ideal gases and an idealized intermolecular-force potential. This formula, often called Sutherland's law, is still commonly used and most often gives fairly accurate results with an error less than a few percent over a wide range of temperatures. Sutherland's law can be expressed as:
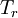 is a reference temperature.
is a reference temperature.
 is the viscosity at the
is the viscosity at the 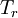 reference temperature
reference temperature
- S is the Sutherland temperature
Some authors instead express Sutherland's law in the following form:
Comparing the formulas above the 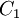 constant can be written as:
constant can be written as:
References
- Sutherland, W. (1893), "The viscosity of gases and molecular force", Philosophical Magazine, S. 5, 36, pp. 507-531 (1893).